Moscow claims lead in vaccine hunt

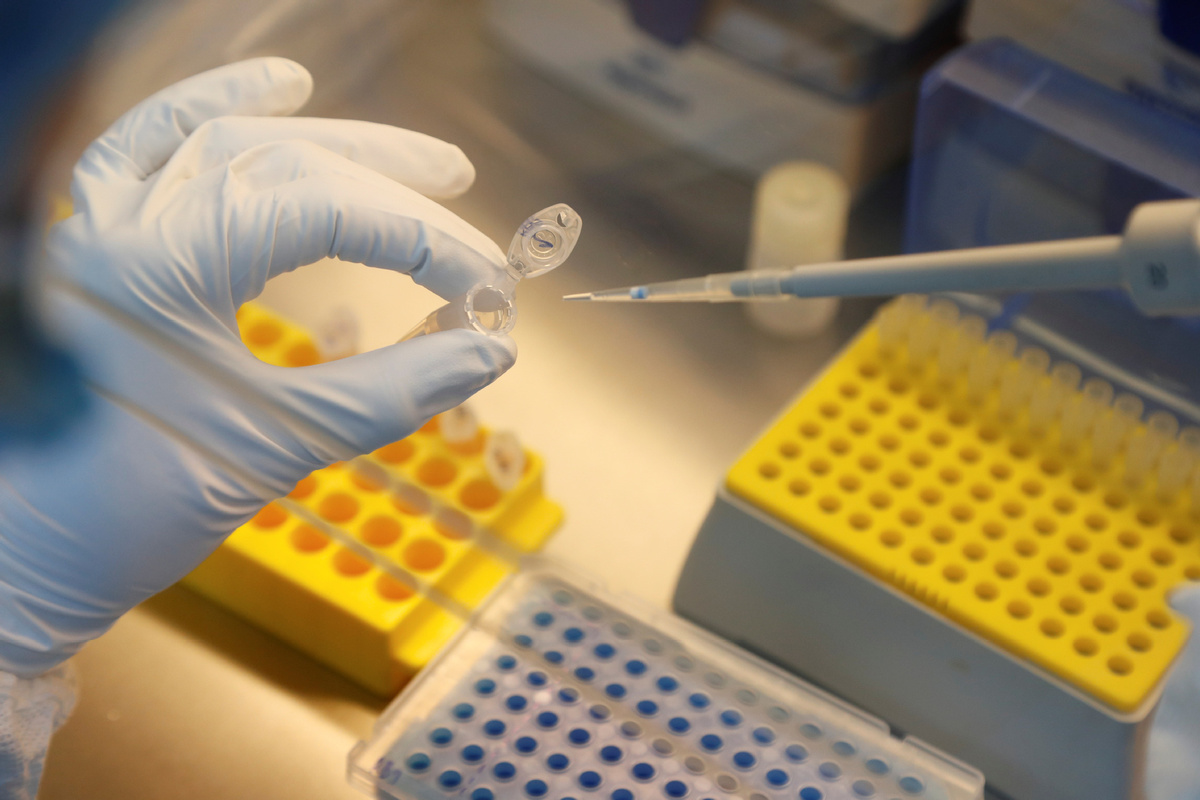
Russian medical researchers have claimed the lead in the race to develop a vaccine for COVID-19.
Media reports say that two vaccine candidates have completed the second phase of trials-the last of which were on humans-and will be made available, initially, to a limited number of people who step forward for injections next month.
While the reports refer to the vaccines as having completed all trials, a leading researcher behind the project said their limited public distribution-slated for Aug 14-15-is the equivalent of a phase III trial.
Three entities, including two Chinese companies, have entered phase III trials-or the final stage of human testing-for their vaccines, Reuters earlier reported.
In Russia, the two vaccines have been developed by the Gamaleya National Research Center for Epidemiology and Microbiology in Moscow. The Moscow center was given the go-ahead from authorities for the latest round of clinical trials that began on June 18.
With the trials completed, Alexander Gintsburg, the director of the Gamaleya research center, said a small amount of the vaccines will be made available to ordinary Russians over Aug 14-15-likening this development, with the follow-up monitoring, to a phase III trial.
The first vaccine, in the form of a solution for intramuscular administration, was put through trials at the Burdenko Military Hospital.
At the same time, the second candidate vaccine-in the form of a powder prepared as a solution for intramuscular administration, was given to 18 volunteers at Sechenov First Moscow State Medical University.
Gintsburg said the administration of doses to those who come forward next month would proceed under strict supervision.
Early results from the first small-scale human trial of the vaccines developed by the Gamaleya research center had shown them to be safe, according to a report by the RIA news agency on Sunday.

Treatment might be free
It's envisaged that the vaccines will be provided to the public in designated medical institutions and, possibly for free, with the expense borne by the government.
Private companies would begin mass production of the vaccines by September, Gintsburg said of the envisaged timeline.
In the human trials at the Sechenov First Moscow State Medical University that began last month, nine of the 18 volunteers received one dose of vaccine, and the other half were given the prospective booster dose to test.
"The research has been completed and it proved that the vaccine is safe," said Yelena Smolyarchuk, the head of Sechenov University's clinical research center.
Each group of the volunteers will be discharged from the Sechenov University facility, on Wednesday and next Monday, after spending 28 days in isolation to guard against exposure to other infections. The volunteers, aged 18 to 65, will be monitored for a further six months after their release.
"Data currently available... shows the volunteers have developed an immune response to the coronavirus vaccine," the Russian Defense Ministry, which is involved in the trials, said on Monday. It added that a further 20 volunteers were administered one of the vaccines at a military hospital on June 23.
The decision to involve the general public in vaccine trials underscores Russia's desire to move ahead fast with its testing of the sought-after coronavirus vaccine, said the Moscow Times.